
A new model of chemical order was the result of an ongoing collaboration between chemical and physical research projects with evolving aims and standards. This decoupling of elemental identity from weight allowed for a coherent concept of isotopy, which arose from investigations on the decomposition series of radioelements and their unstable chemical identity. 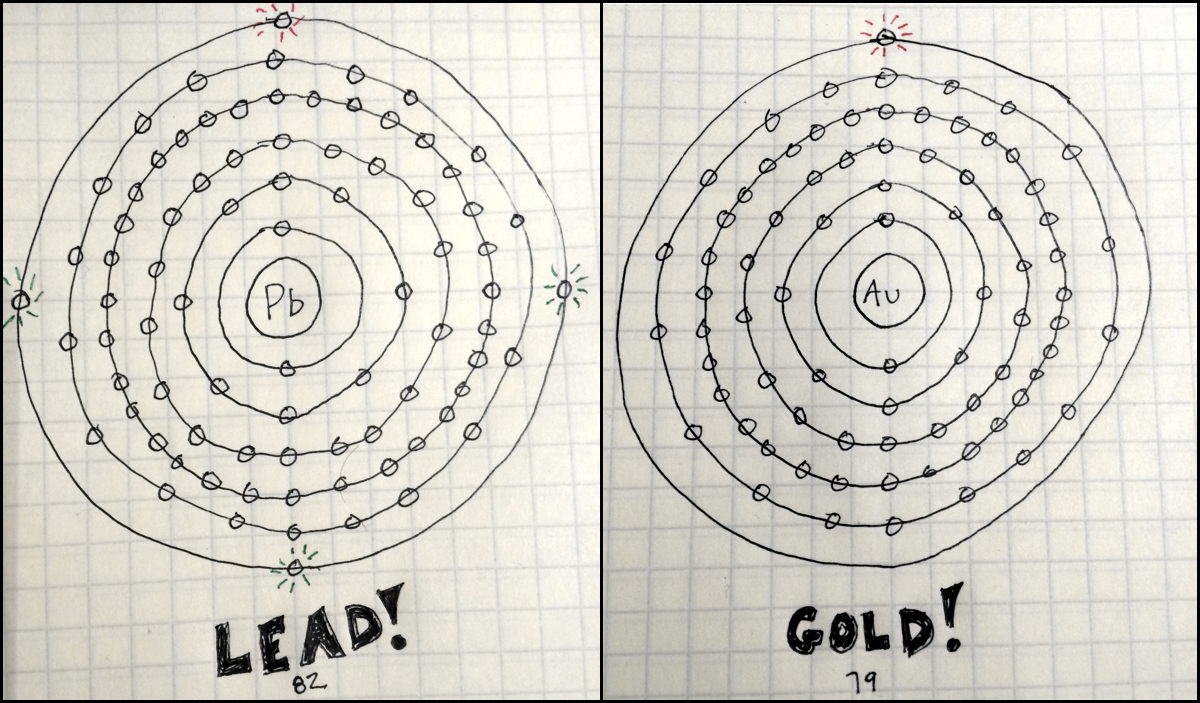
Then, in the second decade of the twentieth century, developments in structural theories of the atom and the observation of regularities in radiation-scattering experiments and X-ray spectra led to this concept being mapped onto nuclear charge (and, later, the structure of the nucleus). Focusing primarily on research by Frederick Soddy (1877–1956) and Ernest Rutherford (1871–1937), we show that, in the context of differentiating disciplinary projects, the adoption of a complex and shifting concept of elemental identity and the ordering role and rule of the periodic table led to a relatively coherent notion of atomic number. We provide a detailed history of the concepts of atomic number and isotopy, from 1896 to 1914 (before the discovery of protons and neutrons), that draws attention to the role of evolving interplays of multiple aims and criteria in chemical and physical research. It is an episode of scientific collaboration and partial integration without simple, wholesale gestalt switches or chemical revolutions. After key concepts were considered resolved and their territories were clarified, chemistry and physics resumed autonomous projects, yet remained bound by newly accepted explanatory relations. This concept received motivation from empirical investigations on the decomposition series of radioelements and their unstable chemical identity. Subsequent attention to valency, still neglected in the secondary literature, and to nuclear charge led to a decoupling of the concepts of elemental identity and weight and allowed for a coherent concept of isotopy. Focusing on research by Frederick Soddy and Ernest Rutherford, we show that, in the context of differentiating disciplinary projects, the adoption of a complex and shifting concept of elemental identity and the ordering role of the periodic table led to a relatively coherent notion of atomic number. Students may be interested in the even smaller particles of quantum physics-like quarks, bosons, neutrinos, and antineutrinos.We provide a detailed history of the concepts of atomic number and isotopy before the discovery of protons and neutrons that draws attention to the role of evolving interplays of multiple aims and criteria in chemical and physical research. They are intended to get players thinking about what they already know about elements. General questions about the properties of elements assume standard temperature and pressure (helium is liquid below -268☌ and gold is a liquid above 1064 ☌). For this game, the most common isotopes of the chemical elements are used. 
This game presents a simple introduction to the Rutherford-Bohr model of the atom and the way we organize the elements. NS 5.8.2 - Develop an understanding of properties of matter Note:
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
